Streptomycin, an aminoglycoside antibiotic produced by Streptomyces, is closely related in structure, pharmacokinetics, and antimicrobial activity to dihydrostreptomycin. These compounds are widely used in veterinary medicine to treat bacterial infections in livestock such as cattle, pigs, sheep, and poultry, where they are effective against a range of pathogenic bacteria.
In beekeeping, regulatory frameworks differ by region. Streptomycin is prohibited in European apiculture due to food safety concerns, while in the United States it is permitted for specific disease outbreaks, such as American foulbrood, a serious and highly contagious bacterial infection affecting honeybee colonies. Even where its use is authorised, strict controls apply, including a mandatory withdrawal period—typically around four weeks—before honey can be harvested for human consumption, to minimise the risk of residue contamination.
Despite these measures, international regulations generally prohibit the presence of antibiotic residues in honey intended for the food chain. As a result, reliable and highly sensitive analytical testing is essential to ensure compliance and protect consumer health.
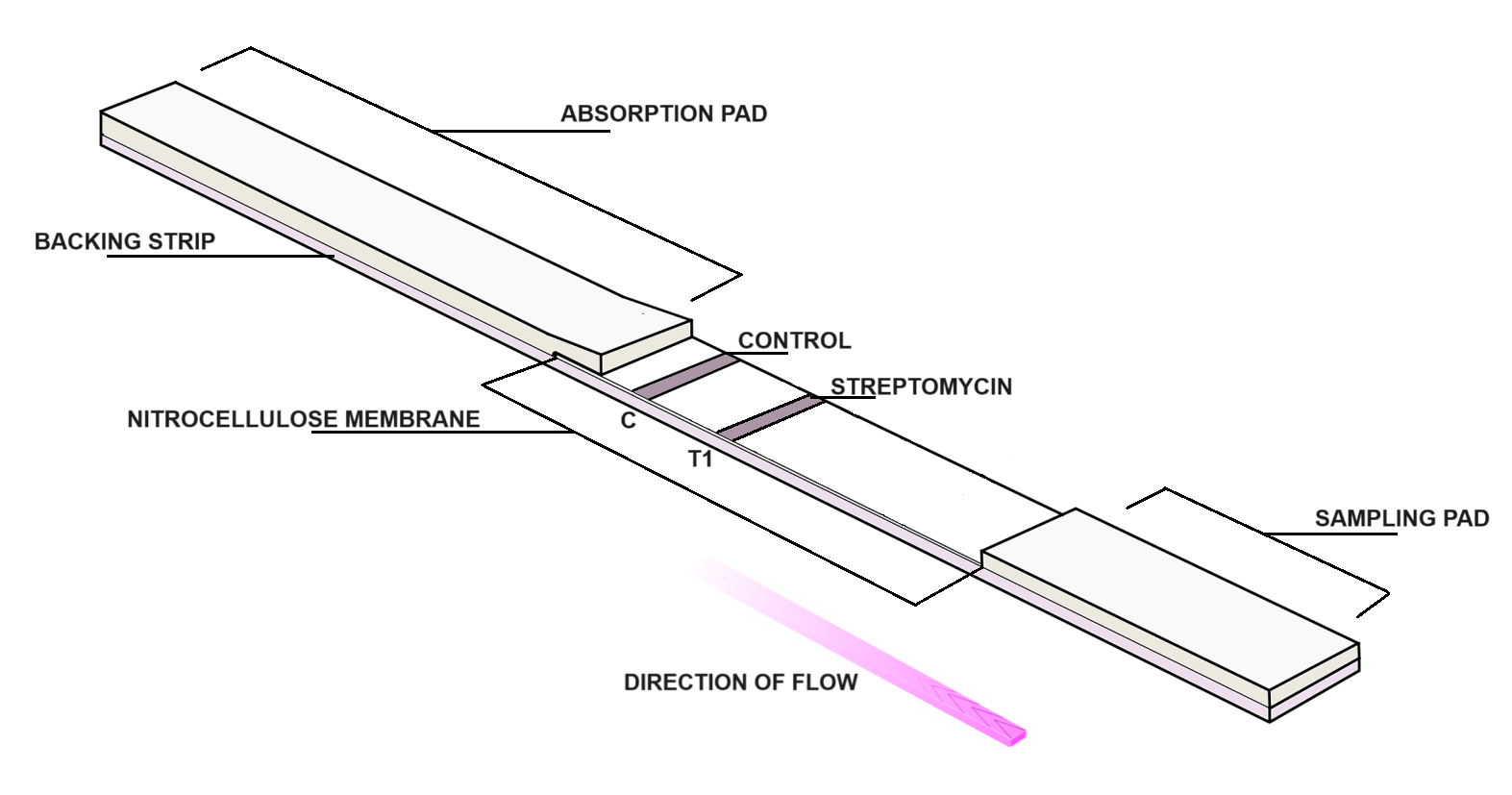
Streptomycin RAPID has to feature as part of this quality control approach, providing a fast and effective screening solution for the detection of streptomycin residues in honey. Rapid testing enables laboratories and producers to quickly identify potential contamination, support regulatory compliance, and reduce turnaround times compared to conventional analytical methods.
The detection of streptomycin residues is critical, as their presence may contribute to allergic reactions in sensitive individuals and further exacerbate the global issue of antimicrobial resistance. Therefore, robust monitoring systems, including tools such as Streptomycin RAPID, are essential for safeguarding food safety, maintaining product integrity, and ensuring consumer confidence in honey products.